Pushing boundaries to improve product performance and patient care
Why?
That was the essence of the questions Tony Pearson-Foldenauer kept asking himself in the hours and days after his father-in-law suffered a heart attack within minutes of having a (non-Cook) stent placed in January 2020.
“Originally, I was concerned about vascular dissection,” Tony said. “But they went in and put a second stent in and everything’s fine—he’s doing great.”
But Tony, who has a background in risk management and quality, needed to know more. How often was this sort of incident occurring when this stent was used? Were there better options for his father-in-law of which the family wasn’t aware?
“I investigated a little bit further to understand the performance of the device overall,” Tony said. “What I found was somewhat intriguing. From the standpoint of the device he had implanted—it was actually state-of-the-art. There were other devices on the market that were more prone to vascular dissection than the one that he had implanted.”
In doing this research, he also realized something else. There is information out there—and a methodology that could be developed using that information—that could help Cook detect and address issues with our own devices much earlier and effectively than ever before. It would also give Cook a better idea of how our devices stood in relation to our competitors in the areas of safety and performance.
It was a textbook example of taking existing tools, and by being relentlessly inventive, finding new ways to use them.
“A-ha” moment
Tony’s detective work required a bit of role playing—he looked at all the publicly available device information through the lens of the people who collect, analyze, and share that data—the regulators.
What he found was the FDA and European regulators were looking at device incidents from different angles. The FDA keeps track of the total number of incidents per device, while in Europe, the documented number is the rate of occurrence—the number of incidents divided by the number of that device that were used. A device may have what seems to be a high number of incidents, but if it is a device that is used in a large number of procedures, the actual percentage of incidents may be relatively low compared to a less-frequently used device with the same number of incidents. Tony realized a combination of the two views provided a much fuller picture of device efficacy.
It was an “A-ha” moment for Tony.
He started taking the number of Cook device incidents recorded in the FDA’s MAUDE (Manufacturer and User Facility Device Experience) database, which collects device incidents from around the globe—and filtering it through the European methodology—incidents per number of devices sold.
Tony also realized that that same methodology could help Cook identify issues with a product before the device ever left manufacturing.
PPA
The result of his research is a product performance assessment (PPA) methodology that is being gradually introduced into some of the Cook Inc. manufacturing areas.
“Our main focus for the first half of this year was utilizing the PPA in remediation at Cook Inc.,” said Devon Cassidy, senior manager, Quality Engineering and project lead for the implementation of the PPA. “But it’s really about ensuring patient safety and not just making sure we are hitting the mark on compliance. Let’s focus on what matters—making sure that our product is safe.”
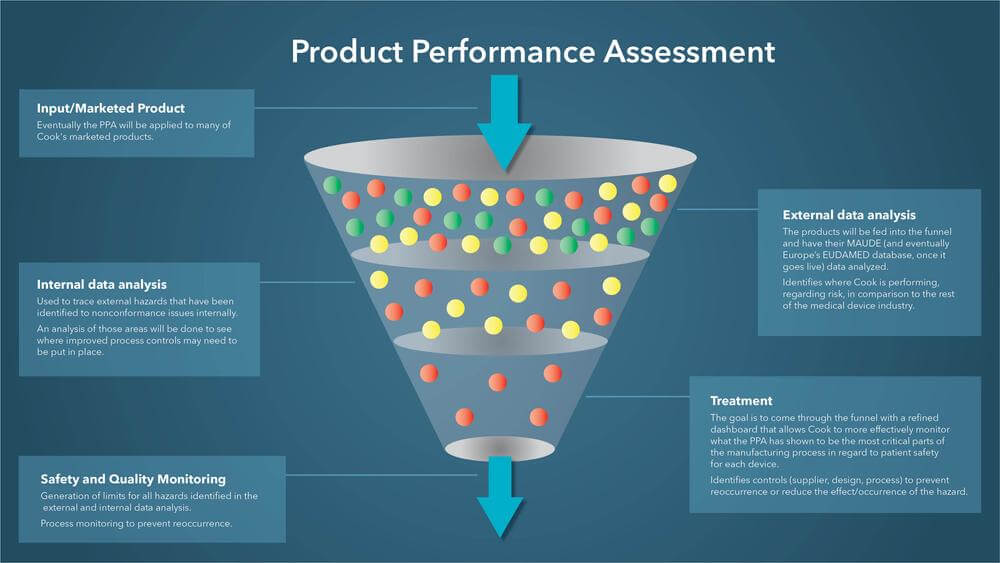
The hope is that eventually the PPA will be applied to many of Cook’s marketed products, a process that Tony likens to running them through a funnel. The products will be fed into the funnel and have their MAUDE (and eventually Europe’s EUDAMED database, once it goes live) data analyzed. That information will be used to do an internal data analysis to determine where in the manufacturing process there may be correctable issues. Then an analysis of those areas will be done to see where improved process controls may need to be put in place.
The goal is to come through the funnel with a refined dashboard that allows Cook to more effectively monitor what the PPA has shown to be the most critical parts of the manufacturing process in regard to patient safety for each device.
“It’s a picture of the device that enables us to identify where the device is failing, what is failing, and how many times it’s failing,” Tony said. “This, in conjunction with some of our internal reports, enables us to link the hazards that we see from the field to our internal nonconformance codes. That’s highly critical for us because that enables us to start trending those nonconformance codes to ensure that we don’t see these issues creep up again.”
The PPA will also give us some insight into how our devices are performing versus our competitors.
And it shouldn’t come as a surprise that this additional data will allow Cook to operate more efficiently and, thus, see some cost savings—one of the major areas of focus in the Mid-Range Master Plan.
In the early going, Devon and her team have identified several ways in which the PPA findings can have a positive financial impact for Cook.
These include:
– Product transfers from one manufacturing facility to another.
– Elimination of inspections that aren’t directly linked to product performance and safety.
– Identification of each device’s critical to quality (CTQs) elements.
“This tool allows us to identify what’s really important to the product and what inspections are important to the product,” Devon said.
Promoting safety and performance for physicians and patients
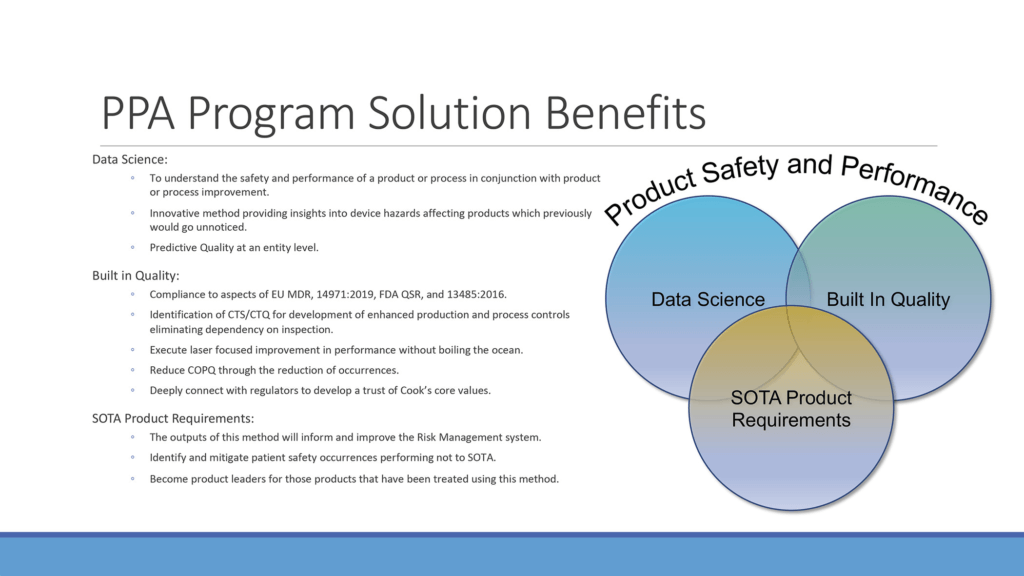
Robert Popp, vice president, Quality, Cook Medical and executive sponsor for the PPA program is excited about the potential of the methodology. He knows, though, that excitement must be tempered not just by a thorough vetting of the tool, but also an accurate analysis of the data it produces. To that end, Cook’s Business Insights team and CMH Statistics team has been heavily involved in the PPA’s development.
“We want to make sure we aren’t seeing false signals—making sure that the information we’re seeing is indeed true activity,” Robert said. “It takes us some time to validate. We don’t want to misinterpret the data. We want to make sure that we learn how to read the data right. We want to improve the way we’re assessing the risk of safety-related events to our consumers. That’s the main objective here. We believe this is better than the current methods used for assessing overall risk to patients.”
He envisions the PPA eventually being used throughout Cook’s entire device portfolio—and beyond.
“This methodology—it’s unpatented—it can’t really be protected, it’s drawing from a public database,” he said. “We’re using some advanced statistical tools to get to the data—that’s what we would say is proprietary to us. We’re not going to let that secret out, but somebody can figure it out very easily. We’re first to market, but the fact of the matter is I don’t think Cook wants to protect something that promotes safety and performance of products. We want to say this is what we’re doing, we stand behind this methodology, and looking at our products moving forward this is the way we’re going to assess that our products result in the safest outcomes that are available? That’s the mentality we want. We would like to publicize this. We would like to go out and promote our innovative approach.
“The next guy is going to come along and duplicate what we do and the world would be a better place—and I hope Cook gets recognized for being the leader beginning to foster this way of thinking.”
