Good news in 2016 for Zilver PTX
It is shaping up to be a very happy year already in Peripheral Intervention. While people around the world strive to keep their New Year’s resolutions, our resolve in PI has always remained the same…to help patients suffering from venous and arterial diseases in the periphery receive the best therapies possible.
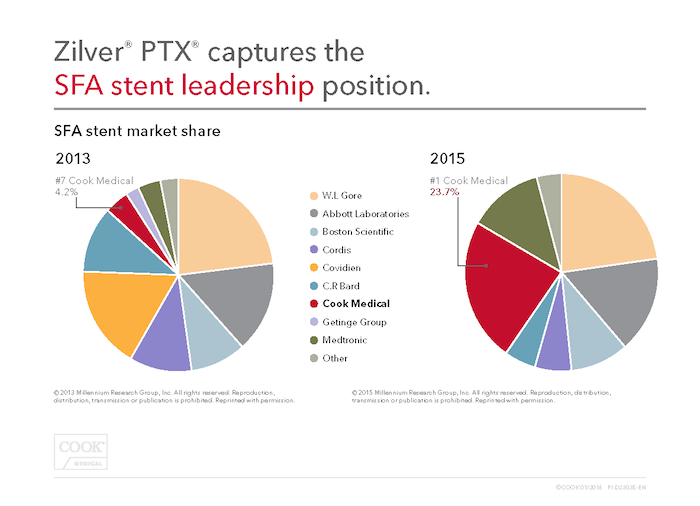
In addition to receiving FDA approval for our new thumbwheel delivery system for Zilver® PTX®, the Millennium Research Group recently announced that Zilver PTX is the 2015 U.S. market share leader for stents in the superficial femoral artery (SFA). This means only one thing to us: more patients diagnosed with peripheral arterial disease (PAD) are being treated with this proven, long-lasting therapy.
PAD is a common circulatory problem that causes blood vessels to narrow or become clogged. It is most often caused by smoking, diabetes, obesity, and genetic predisposition.
More than 200,000 patients in 48 countries have been treated with Zilver PTX, the world’s first and only drug-eluting stent approved to treat PAD in the SFA. Given the fact that we have the only 5-year randomized controlled trial data for SFA stents, it is gratifying to know that physicians also feel confident in this widely tested and adopted therapy. (Find out how drug elution works.)
After receiving FDA approval for Zilver PTX in 2012, the market share uptake has been astounding. In 2013, it picked up 4.2% of the U.S. SFA stent market share (#7 among competitors). Only two years later, Zilver PTX captured 23.7% of the market share, leading all U.S. SFA stent producers. There are a lot of choices out there to treat PAD in the SFA, so it’s great to see physicians reach for Zilver PTX as their stent of choice.
Zilver PTX deployment is now simpler than ever with our new rotating thumbwheel delivery system. (Watch the video below to see the thumbwheel in action.) Cook engineers were tasked to develop this easy-to-use handle to improve accuracy and deliverability on our previous pin-and-pull system. The new Zilver PTX system also includes 120 mm stents for longer lesions as well as a 25% smaller, streamlined box to save room on cath lab shelves.
We are not stopping there. We have several more projects in the pipeline that will continue to help patients diagnosed with PAD.
Thank you to all who have worked so tirelessly to make Zilver PTX the new market share leader as well as bring to market the new Zilver PTX thumbwheel delivery system, 120 mm stents, and streamlined packaging. Cheers to your invaluable efforts! And Happy New Year!

Great and improved design feature with the ‘scroll wheel’ deployment!
Great way to welcome in Spring 2016!
Congratulations, PI on all of your hard work! This is fantastic news!
Very impressive and accurate placement!
Wow……impressive ! Congratulation to all ZPTX team!
Very cool! Love to see Cook continuing to evolve and capture this market. My mom has some of these stents so I’ll look forward to hearing the feedback from her doctor when I see him!
This is really cool Mark!!! Thanks for sharing. We are excited about Zilver PTX clinical study going well in China. Hope more Chinese patients are treated by Zilver PTX soon!!!
wow, it’s cool to see it in action! Almost makes it look easy
This is bigger than I imagined. My customers are receiving the new delivery with open arms! Game changer for us
New definition to March Madness with the release of the Thumbwheel. Thank you ZPTX team!
Hooray!