Hemospray’s epic journey to the US

Cheers are erupting throughout Cook. Can you hear them? It’s been a long road for the Food and Drug Administration (FDA) to grant permission to market Hemospray® Endoscopic Hemostat in the US, but a dedicated team of regulatory specialists, managers, leaders, clinical experts, engineers, and more crossed the finish line this week.
The journey began in May 2010, progressing with many twists and turns. Along the way, Cook was perfecting clinical studies, creating marketing plans, and designing training programs surrounding the release of Hemospray. Now we are excited and ready to make this important treatment option available to patients.
For readers who are not familiar with the FDA process and lingo, read this helpful list of definitions for FDA terms and acronyms.
What is Hemospray?
Hemospray is a single-use device containing an inorganic, hemostatic powder used by clinicians during endoscopic treatments to stop non-variceal bleeding in the gastrointestinal (GI) tract. The powder acts as a cohesive and adhesive mechanical barrier that absorbs water and forms a gel that covers the bleeding area. The powder is propelled with carbon dioxide (CO2) toward the bleeding site through the channel of a catheter attached to a single-use device.
Ironically, Hemospray has been manufactured for years at Cook Winston-Salem, yet the product was unable to be sold and distributed within the US. The Hemospray device has already been registered for use in upper GI bleeding in the European Union, Australia, and Brazil, and for use in lower GI bleeding in Canada, Mexico, and Hong Kong.
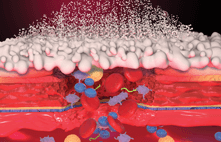
“With FDA’s permission to market, the division may elect to begin registering the product in yet more countries,” explained Marge Walls-Walker, senior regulatory specialist at Cook Winston-Salem. “Hemospray meets an unmet need in the array of endoscopy GI bleeding treatments around the world,” added Marge.
Regulatory strategy: unusual turning points, teamwork, and two mineralogists
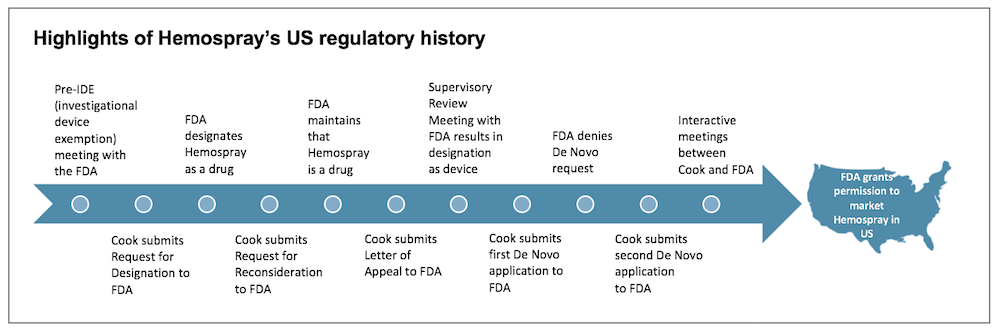
An important part of the FDA product submission process is initial determination of the class of the device and thereby the regulatory strategy. “Our initial meeting with the FDA in 2010 did not go as we had hoped; the FDA considered Hemospray a Class III device, which meant considerable time, money, and clinical trials to get to market,” said Marge. “The team stepped back to reconsider options. Ultimately, Bill Voorhees, vice president and chief science officer at MED Institute who has helped guide Hemospray through the process since the first meeting with the FDA, reached out to determine if the FDA would reconsider the Class III designation. The FDA’s response was to suggest that we submit an RFD (Request for Designation) to the agency to determine the path to market, and classification.”
After submission of the RFD, the team was hit with a surprise. The FDA came back with a completely different determination: they designated Hemospray a drug instead of a medical device. This drug designation led to three years of negotiation with the FDA, filled with appeals, written statements, review meetings, and bringing in outside expertise to prove the product was indeed a medical device.
Research & Development Engineer Shaun Gittard gathered outside experts to answer the FDA’s questions. Hemospray’s panel of outside experts included two mineralogists, two physicians, and one hematologist. “It is not typical to bring in outside experts who are not physicians during the FDA process,” explained Shaun. “However, because of the nature of the questions about how the powder works and interacts with the body, we invited the mineralogists and hematologist to share their knowledge.” Fortunately, after hearing the experts at the appeal, an FDA commissioner declared the product a device.
Double De Novo
Another unusual step in the Hemospray FDA process was the double De Novo requests. (De Novo is defined in our FDA Glossary.) The team requested a direct De Novo from the FDA for the Hemospray device. The team’s first request to classify Hemospray as a De Novo device was denied, though the FDA said they would consider a new De Novo request if additional information and a summary of the clinical data were provided.
Marge described the process: “We were able to negotiate a meeting with the FDA review team to discuss the steps forward. We submitted the second request, responding to specific questions and asking that any additional questions be dealt with on an interactive basis. The FDA obliged, and after a series of interactive review teleconferences with the FDA to answer additional questions, complete additional testing, discuss mineral science, and make labeling changes, the FDA has now granted permission to market Hemospray, which is the first De Novo product in Cook’s history. Getting Hemospray to this point has required input from virtually every discipline and department within Cook, and the rapid responses to requests for assistance have been truly remarkable.”
The team in Winston-Salem was overjoyed to reach their goal of getting US physicians and patients access to this product.
 The clinical strategy
The clinical strategy
As the Regulatory and Engineering teams focused on convincing the FDA to grant permission to market Hemospray in the US, the Clinical Affairs team developed and initiated the collection of clinical data to support outside US (OUS) registrations and build a safety and performance profile of Hemospray in clinical use. David Wagner, manager of Clinical Affairs at Cook Winston-Salem, has been involved in the clinical study process for Hemospray since the beginning.
The range of GI bleeds is so broad, it is impossible to study all of them. So, for one of the studies, the team chose the fairly common peptic ulcer bleed. After rigorous recruitment and analysis, David and the team at Cook Research Inc. finally got enough data from 67 patients. They named the clinical trial Hemostasis of Active Gastrointestinal (GI) Luminal Tract Bleeding or the HALT Study. “This study was a significant milepost for us. The study represents the largest prospective series of patients studied and treated with Hemospray for peptic ulcer bleeds in the world right now,” David added.
Another study that Cook conducted demonstrated the use of Hemospray in lower GI bleeding, such as for bleeding after the removal of a polyp during a colonoscopy or bleeding caused by cancer in the colon. Cook also helped fund a physician-driven, registry study that includes patients with many types of bleeds. This study will allow us to see where Hemospray works best in the “real world” and identify best practices.
Marketing the new product story
There is more to the story than the FDA granting permission to market Hemospray. Now it’s up to the global brand marketing team to introduce Hemospray to customers with appropriate messaging and well-trained sales representatives who can teach customers how to use the product.
“Greater opportunities for Hemospray are with smaller, community hospitals where they often don’t have the staff available to treat severe bleeds,” said John Devlin, manager of global brand marketing, Endoscopy. “Our goal is to help everyone understand how Hemospray works. We also need to create a clear awareness of the product.”
Team’s persistence pays off for patients
When some of the Regulatory team members were asked why they stuck with the lengthy challenge to get the FDA to grant permission to market Hemospray in the US, here’s what they said:
“I knew it was going to work and be a very good treatment. Physicians were asking for it. They would say things like: ‘If I had this device, I could have stopped the bleeding.’ I was committed to the project because Hemospray can change the course of treatment for a patient. It clearly defines a need, physicians want it, and I saw that the product worked.”
–David Wagner, Manager, Clinical Affairs, Cook Winston-Salem“For me, the challenge of ‘winning the day’ kept me going. Every year, sales increase for this product around the world. Physicians have even contacted the FDA to push Hemospray through.”
–Marge Walls-Walker, Senior Regulatory Specialist, Cook Winston-Salem“I knew Hemospray was a great product the first time I saw it used. Its acceptance and success overseas proved it. ”
–Bill Voorhees, Vice President and Chief Science Officer, MED Institute
Congratulations to each team member whose dedication and perseverance has brought Hemospray to the US market! Read the press release on our US website.
AVAILABILITY AND INDICATIONS FOR USE
At the time of this blog’s publication, Hemospray Endoscopic Hemostat has been authorized by regulatory bodies to use for hemostasis of nonvariceal gastrointestinal bleeding in the US, Canada, Mexico, and Hong Kong. In the European Union, Australia, New Zealand, Egypt, and Brazil, Hemospray Endoscopic Hemostat is authorized to use for hemostasis of nonvariceal upper gastrointestinal bleeding.
this story demonstrates what persistence means… nice work all!
What a great story – nice work! Congratulations to the team and to Cook…
Congratulations to the team! My first experience with and exposure to Cook was with the Hemospray HALT study. Glad to see this will finally be available in the US!
Congratulations! Great work with perseverance and dedication!
Great product development-Congratulations to all.
Improving patient care and saving lives; our story!
Endurance, persistence and dedication are leading qualities in any game plan……….they truly paid off in this plan. Congratulations to each one on a job done so well.
Congratulations to all of you! What a hard work and now such a deserved success. Surgeons and patients will appreciate it!
This is a great story of dogged persistence and for the right reason- better patient care- two things that are deeply rooted in our culture. Thank you Mr. Cook for making us who we are!
This is wonderful. Congratulations to everyone on this accomplishment!
Great persistence and fascinating journey! Congrats to all involved!
Our history suggests that when we believe we have an idea for better patient care, we stick with it. Well done everyone!
Congratulations to the team–you effort and commitment was above and beyond the norm!
I’m glad we stuck with this and worked with FDA to bring this product to market! A big win for patients.
What a journey! Thank you and congratulations to all involved in bringing this life saving technology to market. This will save lives.
Barry
What a fantastic story about determination! Great job to everyone that worked on this fabulous product!
Congrats all, great accomplishment and persistence.