Earth Day update: new chemical management group
 Happy Earth Day to all Cook employees! We are thrilled to be celebrating our incredible planet with you today. Cook’s global work force is diverse, passionate, and driven to enact positive change. One important way to do this is to integrate sustainability into the fabric of our company and adopt a new lens through which we view our business decisions.
Happy Earth Day to all Cook employees! We are thrilled to be celebrating our incredible planet with you today. Cook’s global work force is diverse, passionate, and driven to enact positive change. One important way to do this is to integrate sustainability into the fabric of our company and adopt a new lens through which we view our business decisions.
Some projects are clearly aimed at improving the environment, such as energy efficiency projects in our facilities, improved recycling programs, or streamlining product shipments to reduce greenhouse gas emissions. Other projects are less clearly related to the environment but are crucial to the longevity – and yes, sustainability – of Cook as a company. For example, you might not think of chemical management when you think of our environmental efforts. Understanding the chemicals that exist in our products and processes is an important step to better understanding our impacts on the environment, and ultimately on patients’ well-being.
Chemical management group assembled
Chemical management projects have arisen from regulatory and compliance needs as well as customer requests. Cook recognizes the need for increased visibility and dialogue around chemicals of concern in our products and manufacturing processes. “Chemicals of concern” are those that might have adverse effects on people or the planet. We need to be able to share with our regulatory bodies and customers where these chemicals occur in our products, the concentrations at which they occur, and what kinds of impacts these chemicals might have on patients and the environment.
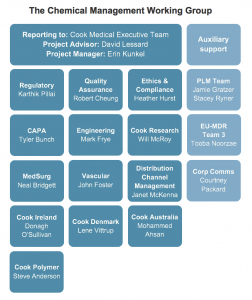
Our new Chemical Management Working Group has brought together individuals from most Cook divisions, departments, and manufacturing locations to create an interdisciplinary dialogue around chemicals in our products and processes.
This group has three main objectives:
- Collection of consistent and controlled chemical data on all our products.
- Development of a database for secure storage and easy reporting on this data.
- Creation of a prioritized list of chemicals.
Working group member Tyler Bunch, a CAPA engineer who works closely with research and development efforts at Cook, said “The work from this group will put us in a better position to meet the needs of customers and patients in regions where we currently can’t comply with their requests for information.”
How does the chemical management project impact Cook?
This project is critical to meeting our high standards for compliance and customer support. We need to understand the ingredients in our products and the impacts these ingredients can have on both patients and the environment over time. Patient well-being will always come first for us. The better we manage the chemicals in our products and processes, the more confidently we can fulfill our mission to provide leadership in pioneering innovative medical solutions to enhance patient care worldwide.
How can you get involved?
If you feel passionate about this project or have an idea that could help us, let us know in the comments or by emailing our sustainability coordinator, Erin Kunkel at erin.walsworth@cookmedical.com.
Awesome! Kudos to the working group members.
Very important, creates trust on all sides!!!
Great initiative !! ….our patients,customers,Cook’s all and our planet will be happy !!
Great to hear as sustainability credentialing is becoming of more importance within commercial tenders within the UK & Ireland
Great and IMPORTANT initiative. Thanks to all
I agree, this is a great initiative, increasing number of customers request chemical data on our products, a database would be a great first step forward. Some countries have clear strategies to ban certain critical chemical from their markets.
I agree with Christa, and would add that this information will also be important in demonstrating biocompatibility of our devices. We are increasingly being asked to carry out chemical characterization as part of ISO standard 10993 (specifically, part 18).
This is an important initiative. Thanks to all the working group members who have stepped to support this work!