FDA audit milestone reached
Earlier this month, an FDA inspector visited P48 to review the progress we’ve made on last fall’s warning letter. Overall, the inspector was positive about the work we have been able to accomplish in such a short time. The FDA is happy to see our commitment of time and resources to the audit response projects.
So, I thought this would be a good time to tell you that phase one of audit work is complete at P48, Canton, and Ellettsville.
Because of the hard work by so many, we finished this work quickly — probably three or four times more quickly than if we had been working at our normal pace.
Global work still underway
As you know, we committed ourselves to the same quality and process improvement initiatives at all the other Cook manufacturing locations. Good progress is being made toward our goal of March 31 completion.
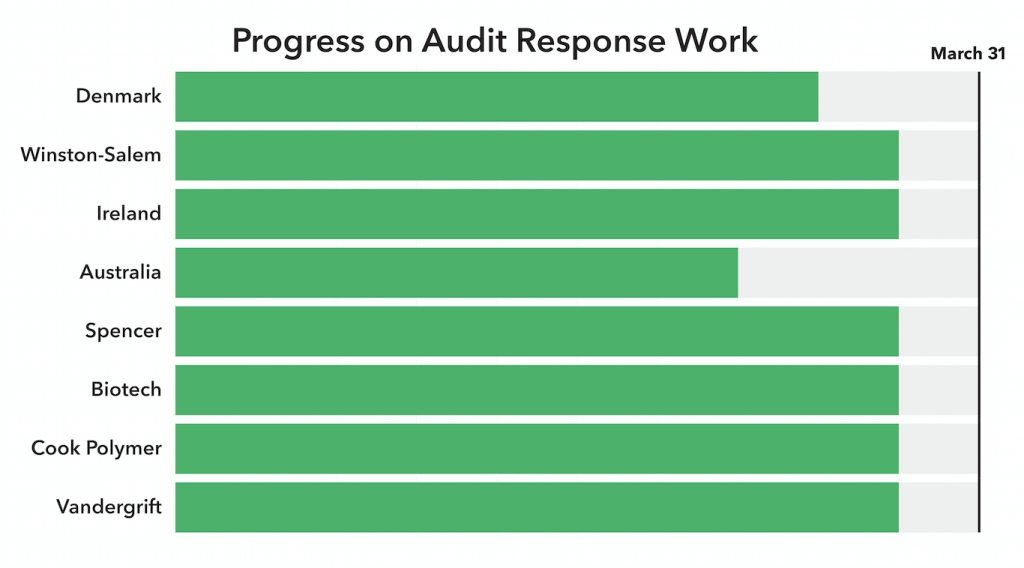 In Denmark and Australia, validations and reporting on a couple of products are taking longer than anticipated. We expect that work will be done in the next few months.
In Denmark and Australia, validations and reporting on a couple of products are taking longer than anticipated. We expect that work will be done in the next few months.
More work ahead
During the re-inspection of the work we did to fix the issues identified in last fall’s warning letter, no problems were found. (Which, if you have experienced an audit, is as good as you can hope for.) The FDA understood that we have not yet had time to fully implement and test our new processes, though, so they are asking us to continue to monitor and improve them.
The inspector will be back to check our progress in six months or so. At that point, if we have addressed the FDA’s concerns and our systems are compliant, we are hopeful that our warning letter from last fall will be lifted so that we can move forward with launching new Class III products (those products that require clinical trials or higher levels of review by the FDA). While a warning letter is in place, the FDA won’t approve new Class III products.
Once the inspector completed a review of our recent work, he moved on to inspect several new areas and processes. At the end of the inspection, he issued a new Form 483 that identified more improvements to be made. A Form 483 is not a warning letter, but it could lead to one. We don’t expect another warning letter, but it is possible.
The new Form 483 included five observations from the inspector:
- Quality Control Inspection: The inspector noted that there are differences in the way some of our quality control employees are doing their work. So we have a team that will be working on reviewing our documentation to make sure it clearly communicates the requirements and steps for final product inspection.
- Process Control in Manufacturing: Again, the inspector visited manufacturing and noticed that there are inconsistencies in the way our employees are approaching their work. In some cases the manufacturing steps aren’t clear or the employee has not been trained in the right way. We’ll be investigating this to determine what we need to do to make sure the processes we use are consistently followed.
- Equipment Maintenance: There are many types of tools and equipment that are used to build our products. We need more-clearly-defined procedures for cleaning and maintaining our equipment. And we need to make sure all equipment is being maintained following those defined procedures.
- Product Complaints: We always work to respond quickly to product complaints, but while we focused resources on the audit response, we developed a backlog of complaints. The FDA took note of that and we have already assigned 16 people from engineering and quality assurance to work on this. We hope to catch up within a month.
- Medical Device Reporting (MDR): The other area noted was reporting of adverse events involving our products. MDRs are not uncommon, but like customer complaints, we need to act on them in a timely manner. For the most part, we do, but three reports (out of 600) slipped through recently that we didn’t report quickly enough. These MDRs often do not mean there was a problem with our product, but it is important that each one is reported to the FDA in a timely fashion.
What’s next
As we’ve mentioned in previous blog posts, we knew that Cook had some work to do to get ahead of current and future regulatory expectations. This process with the FDA has helped us identify areas where we can gain the most ground:
- Training on process — We have grown very rapidly in the last 10 years, but we need to make sure we are keeping up with training the new hires we have. Our trainers need to be experts in training in addition to being experts in the processes they need to teach.
- Clarity of process — We have been creating very specific instructions on all the steps in the production of our products. Now we need to check whether those instructions are clear enough for our team members to follow consistently. Where things are unclear, we need to clarify. Where things are confusing, we need to simplify.
- Monitoring of process — The information we collect from monitoring our processes will help us understand areas that require more training or where we haven’t made the process clear and simple enough to follow consistently. The data we are collecting will also identify the source of problems and help us resolve them.
The FDA is right in expecting us to have clearly defined processes, to train our staff in following those processes consistently, and to have a system that monitors the effectiveness of our processes. It’s important for patient safety as well as our own efficiency.
The new normal
A lot of people have asked when we will get “back to normal.” The short answer is that there’s a NEW “normal.” It looks a little different than the way we’ve worked in the past few years. We’ve always had a focus on product quality, but we’ll be talking a lot more about how we can continually improve our processes and the products we make.
We are also in the process of developing a longer-term plan for our Quality System that will get us ahead of the regulatory expectations. We will keep you posted as those plans progress. It’s possible that there could be more regulatory actions – other inspections, Form 483s, and maybe even warning letters. But, that won’t change our long-term goal of creating a stronger Quality System. We’ll add the resources that we need to get this right.
Thank you for your patience and flexibility.
Wow, this is big success and progress! Thank you for letting us know and thanks to all our colleagues who have been working so hard on it.
Great update ! Thank you for sharing and Congratulation
Thanks for sharing this update. It’s very important to us.
Keep up the good work everyone! Incremental improvement over the long term is a great thing and will make us a better company!
Great update and love all the communication that’s be provided. Thanks to everyone who has put in the hard work to get phase 1 completed. Great to see everyone working together!
Thank you Pete for sharing this great news, A HUGE THANK YOU to everyone that worked tirelessly to reach this milestone!
Congratulations!!! Cook Group I want say Thanks to all of those who have given their best to work through these processes. I understand how important these FDA Audits can be to our company. The results show how everyone has stepped up and gave their best to make Cook Inc a World class business. This shows we are willing to go above and beyond the call off duty to provide life saving medical products.
Thank you Pete for the Update. GREAT news!
Thank you very much for sharing the updates and happy to know about the process also.
Lot of thanks to our team members…..
Pete, great update. Thank you. Not surprising, the effort we are putting out to address this situation. These moments of adversity test us but as a team and COOK family we will prevail in true COOK Medical spirit.
Here’s to onward and upward, and from PI… Good to Great!! Cheers.
Congratulations! Everyone works hard for Cook. Because Cook is our family.
Thanks Pete.
great job guys! you guys earn a lot of respect for the work done in this short period of time. it certainly will be come another milestone in Cook’s long history to come, another challenge mastered….you’d be proud of!
uncountable thanks and we all stand by you!
Congratulations!!! It’s really a big milestone… Thanks for everyone’s untiring effort.
I really enjoy reading the blogs, which provides us transparency and visibility on how FDA thing is going. Thanks for keeping us updated Pete.
Thank you for the update, I want to send out a huge thank you to everyone who is working so hard on this!