FDA audit response status update – December
Hello everyone,
We have made a lot of progress on our FDA Audit response goals, and I wanted to let you know how things are going.
Training
All of our 2,600 manufacturing employees in Bloomington, Spencer, Canton, and Ellettsville have been trained on our new approach to nonconforming product. Manufacturing began tracking nonconforming products in early November.
The reports below include the status numbers from P48, Ellettsville, and Canton.
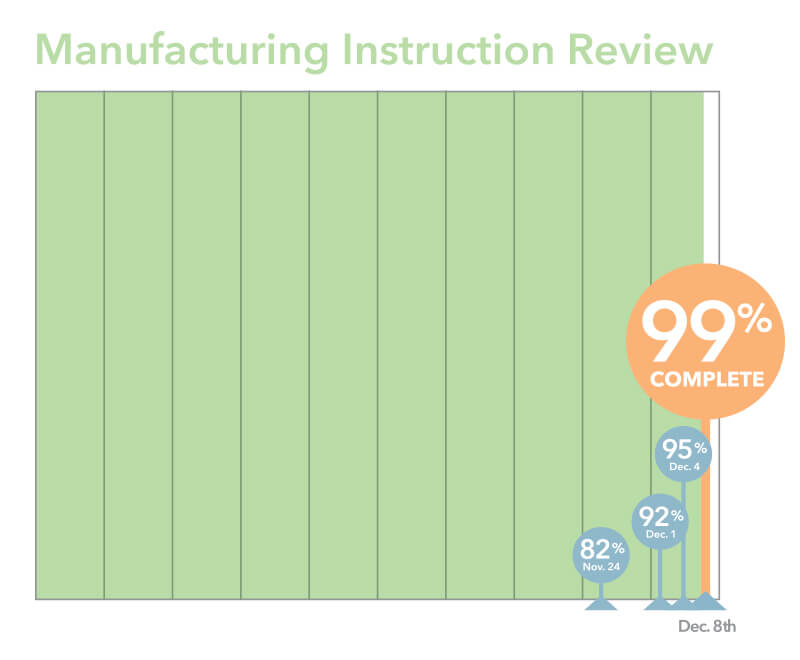
Manufacturing Instruction Review
To make it easier to track and analyze the data from nonconforming product, we needed to more clearly define the processes in our manufacturing instructions. These instructions are the steps that our assemblers follow to build a product. Engineering and production have been working together to review those processes. I’m happy to say that this work is almost complete.
Change Requests
Once manufacturing instructions are reviewed and marked up with changes, the next step is to route and approve those requests. We expect the number of completed change requests to rise quickly now that the manufacturing instruction review is almost complete.
Process Validations
Many of you have commented on the number of validations left to complete. The engineering team has been working since September to validate more than 400 of the processes we use to build our products. It’s a big number, but don’t worry. We started with the processes that are a priority for the FDA, and by the end of the year, 80-90% of those validations will be done. We set the date of December 31 for ourselves, and we are pleased with the progress we’ll be reporting to the FDA at the beginning of January. The remaining validations will be complete before our next report to the FDA in April.
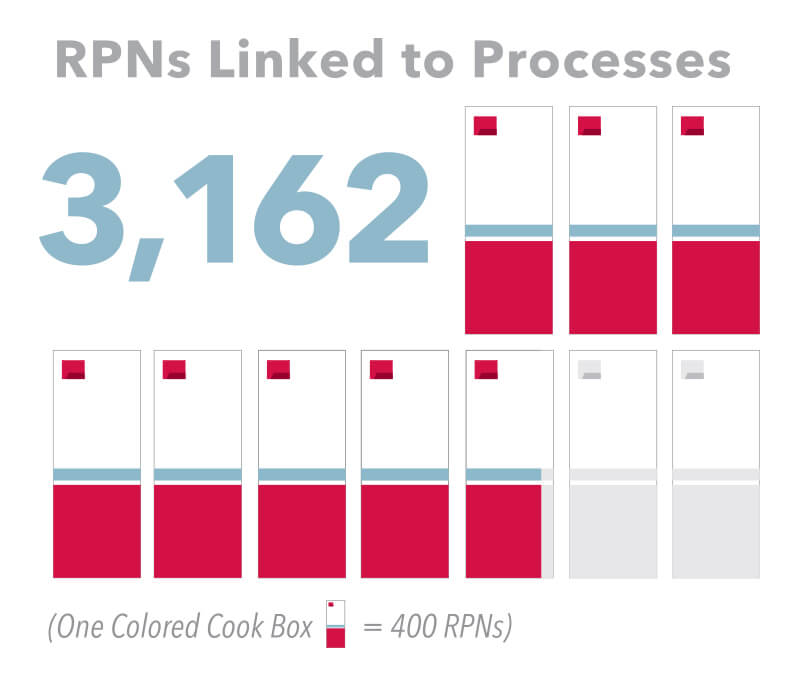
RPNs Linked to Processes
Once the right processes are clearly defined during the manufacturing instruction review, the next step is linking each of our 16,000 product RPNs with the correct process. So work orders will include the process specific to the RPN, and recording nonconforming products will be easy. Production has already started matching up the RPNs to the processes, so you’ll see the number completed grow quickly over the next two weeks.
There has been good progress, and a lot of work will happen in the next few weeks, even over the holidays. I‘ll provide another update in early January. I appreciate the effort and focus that everyone involved has given to this initiative. See this week’s full dashboard report here.
Thank you!
Pete



Impressive progress, and even more impressive, in my eyes, has been the demonstration of transparency, regulatory compliance, pride in quality, concern for the patient, and effective teamwork behind these achievements. I am also really impressed by the overall communication of issues related to the warning letter. Makes me proud to be part of Cook and inspires even more commitment for Cook values and culture.
As Cindy stated so well above, it’s great to see our progress and the level of transparency demonstrated by Cook in sharing the status of our actions. I’m new to Cook and very proud to work for a company that operates as Cook does.
Ditto
This is very impressive! Thank you for these updates.
Well done to all involved
Thanks for the update
Thank you for the update. This is one COOK in action. Great achievements already.
It is impressive how quickly this challenge was addressed and the changes put into practice. Congratulations to all who made this happen!
People pulling together for a common goal…. Cook at it’s best!
Thanks for the updates Pete. Quite like the way you present. We understand it is a challenging process. Once we make it, everyone from Cook shall be proud of it. Look forward to the next update.
Impressive, well done ! Thanks to all involved
Thanks for your update Pete. Incredible to see how we are moving forward towards the goal.
Thankyou for sharing the overall progress!
Go one COOK!
Thank you for the update. Thanks to those all involved.
Thanks for this update. It is very impressive to see how we work on this challenge .
A lot of team effort and coordination going on. This is the way COOK is we all pull together and get done what we need to. Thanks to all involved.
Amazing effort-Cook at it’s best! Well done to all involved.
Nice work!
This speaks a lot for the amount of teamwork and collaboration between the groups that are directly involved.