FDA audit response update — February 2015
For a few months now, you’ve been seeing our FDA audit updates in this blog, in the special editions of Cook News, or on the dashboard posters posted at some facilities. They might look so familiar to you by now that it is hard to tell when a new update comes out.
The numbers in those updates represent a lot of hard work. You probably contributed,
or you helped someone else while they were busy with this effort. Thank you all.
Things are looking good in the latest dashboards.
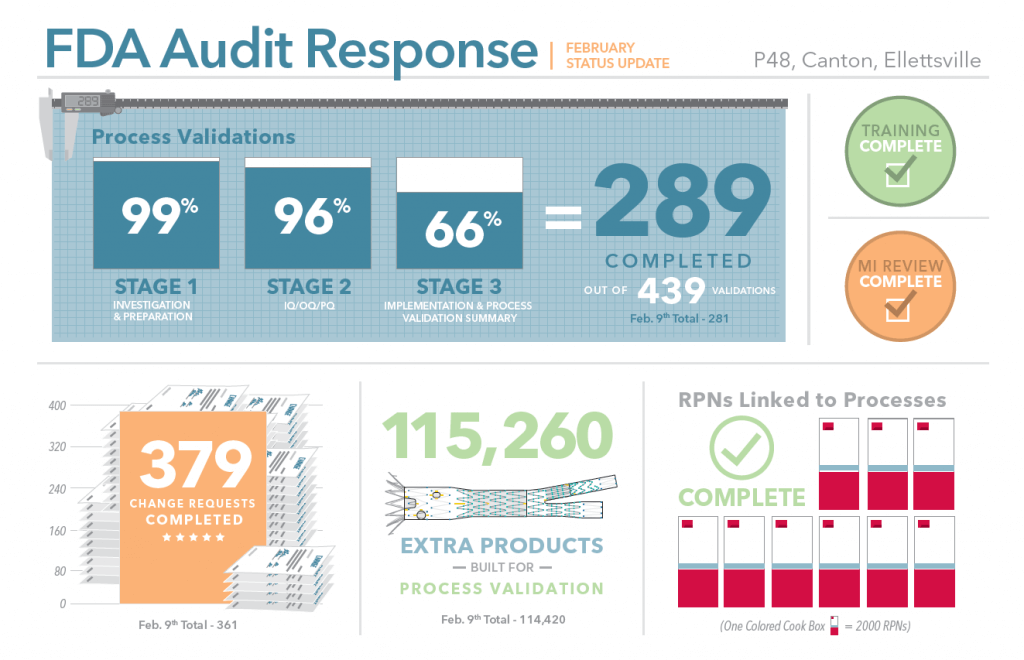
These tasks are complete at P48, Canton, and Ellettsville:

We have trained more than two thousand manufacturing employees on our new approach to nonconforming products. Tracking non-conforming products during production started in early November 2014.

Production, engineering, and IT have worked together to review the manufacturing instructions (MI) for our products. We have found a few additional MIs that needed review, and we are staying on top of those as they come up.

The production, engineering, and IT teams worked through more than 16,000 part numbers (also called RPNs) to match them up with the correct processes and manufacturing instructions. So work orders will include the process specific to the product.
Here’s what we are still working on:
To provide extra products to perform all these validations, while continuing to keep up with customer orders, our manufacturing staff has been working hard. Some departments have even added a second shift. At latest count, 115,260 extra products have been built for use in validations.
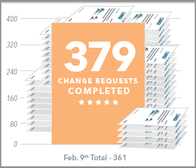
379 change requests have been completed by engineering, document control, quality assurance, and operations. This is in addition to the normal flow of change requests, averaging about 250 approved per month.
The process validations are taking the engineering team a little longer than we expected but we are making good progress. As of February 12, we had completed 289 out of 439 validations. That “completed” number nearly tripled in the last two weeks, so it’s clear we are heading in the right direction. I am confident this step will be wrapped up well before our next report to the FDA in April.
Around the Cook world
The other Cook manufacturing facilities around the globe are well into working on the same tasks—training staff, reviewing manufacturing instructions, linking RPNs to processes, building extra product to validate processes, and completing change requests. If you are located at one of our global sites, you might have seen dashboard posters tracking the local progress.
Reports to the FDA
In early January we submitted a report to the FDA on the seven issues identified in the warning letter. The FDA confirmed that they received our report, stating, “We appreciate your firm’s efforts in correcting Warning Letter issues #3, #4, and #6.” Three down, four to go.
We are on track to have the remaining items completed for our next report on April 8, 2015. While we feel that we have done an excellent job addressing the FDA’s concerns, it’s always possible that they could ask us to do more. If they do, we will jump on it again.
Then what?
At P48, the dashboard posters will come down after we have addressed all the items in the FDA audit. But that doesn’t mean we are done with making improvements to systems and processes in our company.
We are already starting to look at the new data that manufacturing is collecting. The data will help us identify trends, inconsistencies, and areas where we are not as efficient as we could be.
And we need to get back to being ahead of regulatory expectations rather than trying to catch up to them. To do that, we will begin adding new systems and procedures and reviewing our processes to make sure we are ahead of the game.
We have come a long way since we first started on our audit response back in October 2014. The way everyone has stepped up has been impressive. Thank you to everyone who continues to work hard—this process is valuable to us in many ways.
– Pete


Thanks! In my eyes is very important for each of us to know and to be even more proud about our Company and work. We work on the field to be better every day too!
Good to know that we have completed lots. Thanks to efforts of all of involved.
Well done! …and lets ‘bottle’ all this almost unfathomable and much appreciated teamwork… and continue focusing upon those processes that are good and can be much better.
Lets also simultaneously give mediocrity a big whack and replace it with our new found ZEST for continuous improvement, within the spirit of “Plan, Do, Check… & CHECK again… then ACT with purpose to achieve the intended result”.
Once again thanks to everyone for going the extra mile and supporting those values that we hold so dear!!!
Great to hear and thanks to share !!!
Great to hear!
Cooks family continues to grow and improve!
Thanks for sharing Pete. The update confirms we are moving toward the right direction. Always feel great we are doing the right thing. Really appreciate all the untiring efforts made by Cook people. Look forward to the next update.
Good job to all involved. This has been a tremendous task that everyone has undertaken and a lot of good team work has come as a result; and improvements as well.
It looks like significant progress is being made. Good job everyone. I see P48, Canton, and Ellettsville listed but is there a place to look up Spencer’s dashboard along with other Cook Companies? Again, good job everyone!