I never felt alone

“Throughout the process, though, no matter how difficult it got, I never felt alone,” Frédérique said.
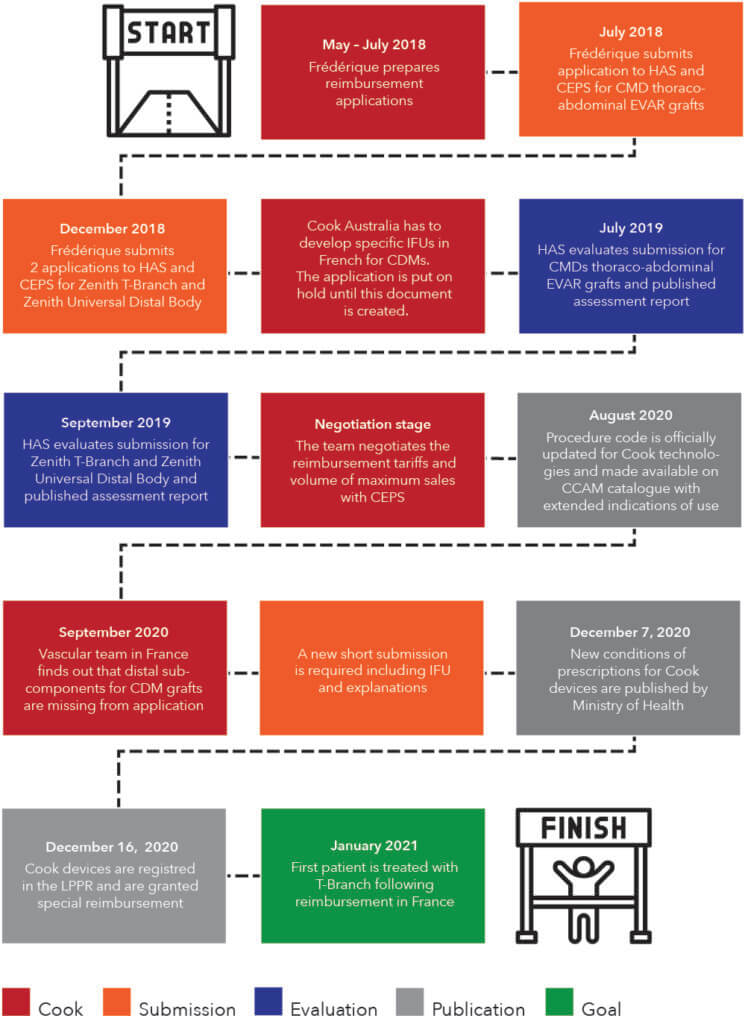
At the beginning of May 2018, she became a Cook employee and covered for Marion Chantegret, a Reimbursement manager in France and Belgium, who was on maternity leave.
“Marion told me not to worry because Mette Agerkvist, our European manager at that time, would help me,” Frédérique said.
 According to Frédérique, she relied on Mette quite a lot in the beginning because of her expertise with the French reimbursement and healthcare system, as well as her knowledge and experience with Cook’s products.
According to Frédérique, she relied on Mette quite a lot in the beginning because of her expertise with the French reimbursement and healthcare system, as well as her knowledge and experience with Cook’s products.
“Each time I needed something, she would find the right information for me,” Frédérique said. “This made my work so much easier, particularly at the beginning when I was new in our company.”
However, Mette went on leave just a few weeks after Frédérique started. Fortunately, she had her colleagues from the European Reimbursement team there to help. At one point, the team consisted solely of Jasmin Wahl-Gravsen (European Reimbursement manager), Vanessa Marenco (a Reimbursement manager in Italy), Jens Johannesen (global director of the Reimbursement function),and Frédérique.
While the team didn’t always have the answers to her questions, they were able to point her toward the people who could help her obtain regulatory documents, vigilance reports, and answers to any technical and clinical data questions on Cook devices.
“Throughout this process, I was grateful for the support I received from all over the company,” Frédérique said. “I did not have to call people back for an answer when I asked them something, which I had experienced at other companies before joining Cook. In general, I can say that I never felt lost or alone in these projects, even if I was at the beginning of my career at Cook.”
Solving problems together
French reimbursement applications can entail a mixture of tight deadlines and solid teamwork among different departments. They must also include a long list of technical, clinical, and economic information on Cook devices. The evaluation process usually takes six months, if nothing is missing.
Frédérique’s field-based colleagues played an important role in the process. The French authorities often require country-specific information, so Frédérique was grateful that she could regularly reach out to Jean-Jacques Chataigner, Vascular business manager for France, and Jean-Philippe Orillard, the advanced clinical support manager.
“They were always supportive. I felt we were not simply working together, there was trust too, and it was a motivating exchange,” Frédérique said. “I never felt stuck thanks to them.”
Another special role in this story was played by several European Vascular colleagues, including John Mogensen and James Guy, who provided her with product information, and Raquel Sanchez, from Clinical Programs, who provided publications on these devices.
Frédérique is field based and has been working from her home office in Rennes (Brittany, western France) since before the COVID-19 pandemic changed conventional work dynamics. In building up the dossiers, Frédérique had to reach out to departments that are far from her home.
“I had to contact many colleagues around the world to get the information I needed to include in the applications,” she said. “However, the support I received from my direct colleagues in our team brought such a relief to me, as there were no opportunities to have physical meetings with the departments I needed to talk to.”
Another challenge that Frédérique ran into was that the required documents needed to be provided in French. However, these devices did not have translated Instructions for Use (IFU). This caused the applications to be put on hold for several months while the team at Cook Australia developed the IFUs, given that those devices are manufactured at Cook Australia.
The first piece of success
In the summer of 2019, the French National Authority for Health, or Haute Autorité de Santé (HAS), released three reports on the devices for which Cook had applied. According to these evaluations, the use of Cook’s thoracoabdominal grafts was allowed, but, due to the complexity of this therapy option, it was limited to certain healthcare facilities.
These limitations had to be included in a new decree before new reimbursement conditions became available, which took time since this restriction is not common. In addition, this all took place in the middle of the COVID-19 pandemic, which, understandably, did not help.
“The reports were just the beginning of a happy ending, though,” Frédérique said.
She and her team had to negotiate the reimbursement tariffs with the Economic Committee for Healthcare Products, or Comité Economique des Produits de Santé (CEPS). They also had to wait until August of 2020 to receive confirmation from the National Health Insurance, or Assurance Maladie, that the indications for this intervention in the relevant procedure catalog (CCAM in French) had been extended to include the thoracoabdominal section of the aorta.
In addition, the HAS assessment report requested that Cook run a post-market study with the devices, as had been done for other aortic endovascular grafts. The results of the study will be needed for the next reimbursement renewal application in five years.
“It is important to start it early in order to not lose any time,” Frédérique said. “Otherwise, we may lose the reimbursement.”
During this process, she worked with Bo Geng, a clinical scientist who involved her in the writing of the protocol for the study and continues to keep her informed of its progress. They both attended a meeting in early 2020 with French health authorities (it including HAS and CEPS) to present the synopsis of the study. The preparation for this meeting, which took place in Paris and was conducted in French, was a collaborative effort. In the end, the meeting went well and Cook’s proposition for a database study was accepted. The next step was the implementation of the study.
Making changes and getting good news
In September 2020, the French Vascular team realized that some of the subcomponents for the CMD grafts had not been included in the initial reimbursement application. Given that some hospitals required the full details to be able to trace all of the parts, the HAS asked Cook for an updated version of the IFU and wanted Frédérique to explain why these parts were not mentioned in the initial dossier.
Cook Australia provided the IFU, and the planning center in London helped Frédérique by giving an example of sizing plans. After several email exchanges, they finally agreed to include the subcomponents in the dossiers.
Finally, toward the end of a surreal 2020, the conditions of prescription and use were published. Cook’s thoracoabdominal grafts were included in the List of the Reimbursed Products and Services (LPPR), were granted add-on reimbursement, and conditions of reimbursement for endovascular grafts were specified. This was very good news just before Christmas.
The first patient
In January 2021, the first patient was treated with a Zenith t-Branch following the approval of the reimbursement in France for this technology.
“We were all extremely pleased when we heard that the first patient case was taking place in Nantes. And I believe this great result was a testament to the commitment we all put in our jobs. Without reimbursement, there would be no patient access to newer technologies in France,” Frédérique said.
Teamwork played an important role in this long and winding road. It is important that those involved feel motivated so that the process does not slow down. Communication was also crucial in this situation as everyone worked on this project from different places around the world. Emails and video meetings—even before COVID-19—helped the colleagues to reach the end result.
“I believe teamwork is important, because we all have our own knowledge, skills, and perceptions,” Frédérique said. “No project can be run alone. Apart from making the project successful, I learned a lot from others, and I hope they also learned a little bit from me. This will make new projects easier. On a personal level, it is also important to be able to communicate with others and to feel that my work is useful.”


Thanks for sharing! Great story!
Thanks, Leigh-Ann!
Well done, great story, thanks for sharing!
Thanks a lot, Elisabeth!
Great story! Teamwork is the key to success! Well done Reimbursement team!
Grazie Sabina!
Great story Vanessa, thanks for sharing and well done Frederique, a real example of how a little help by colleagues can help reaching great results!
Grazie Romina!
Great story….Thank you
Thanks for reading us, Pamela!
Great story! Thanks for sharing!!
Thanks for your comment, Melissa.
Great work Frédérique!
Many thanks Jeannette, I’ll pass on your comment to Frédérique 🙂
Thanks so much for sharing! Great news for patients and a great example of living our values!
You’re very kind, Nicky. I’m glad you enjoyed our story!