OASIS® Extracellular Matrix relaunched in Europe
With Cook Biotech’s relaunch of the OASIS Extracellular Matrix in Europe, ongoing efforts are focused on a clinical trial as well as new marketing and sales pipelines.
Cook Biotech kicks off OASIS clinical trial in UK
Patient enrollment began in a Cook Biotech-sponsored clinical trial (read about that here) aimed at establishing reimbursement for OASIS Extracellular Matrix in the United Kingdom (UK).
Cook Biotech Director of Medical Sciences Jay Hodde said the goal of the study is to obtain clinical evidence that allows Cook Biotech to apply to the UK government for inclusion of OASIS in Drug Tariff. Published monthly by the National Health Service (NHS), Drug Tariff serves as a reference for reimbursement of medical products sold by manufacturers to public hospitals in England and Wales.
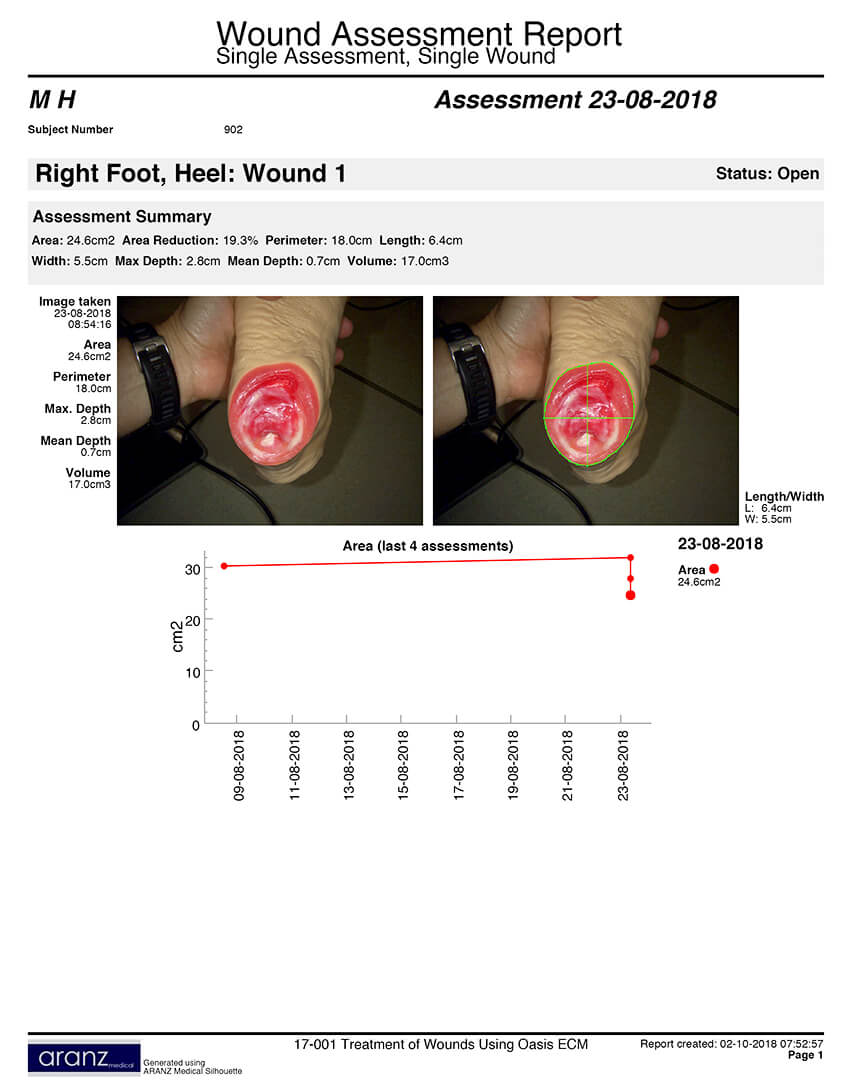
The study will focus on OASIS for treatment of chronic wounds. Patient enrollment is underway at Mid Yorkshire Hospitals NHS Trust and Northumbria Healthcare NHS Foundation Trust. Target enrollment is 40 patients by April 2019. The study will follow patients for up to 16 weeks.
To accurately document patient progress, Cook Biotech purchased three SilhouetteStar™ cameras, which use a non-invasive wound surveillance system for recording wound-healing progress. The cameras record wound-area reduction over time, a metric that is critical when determining reimbursement.
Jesper Thyregod reports on awareness and accessibility of OASIS in Europe
We want to reintroduce OASIS on the European market so that patients in Europe can benefit from this product for wound healing.
So far, we’ve focused on two different areas to create an awareness of OASIS by attending several European congresses. Last month, we were exhibiting at the German Plastic Surgery Meeting in Bochum and the European Burns Association Meeting in Rotterdam.
In the introduction phase, we focus on acute wounds and in patients, hence we are exhibiting at the meetings attended by plastic surgeons. At both meetings, we talked with surgeons from Sweden, UK, Germany, Italy, France, Switzerland, Czech Republic, Romania, China, Afghanistan, and Nepal.
The other area that has our attention is accessibility to customers through sales reps and distributors. We are now in serious negotiations with partners in the UK, Germany, Austria, the Netherlands, Switzerland, Czech Republic, and Denmark, who will be promoting OASIS in their local markets.
At the meetings we have attended in Europe, we have met many surgeons and nurses who are very pleased to see that OASIS is back in Europe. You may say that we are selling “old wine in new bottles.”
Jesper Thyregod is the European Union Business Director for Cook Biotech.
Silhouette Star is a trademark of Entec Health Limited.



SIS heals patients, and can improve their quality of life. Wonderful to see this taking place in Europe. Congrats Biotech!
Thanks Lisa!
My mom was just treated with the extracellular matrix power for a non healing tunnel wound. For 16 weeks the depth only closed by 13% and once the powder was used, the would healed completely in 3 weeks! Truly an amazing product whether it is the powder or the sheets!
Powder that is 🙂
I’m really glad to see this product returned….Years ago one of my co-worker’s son had bed sores very bad. It was a trial, it cleared them up. Great stuff.
AWESOME