Silence is golden
OHNS begins rollout of new Elevo® Snoring Intervention Device
Excessive snoring can damage one’s quality of sleep, energy levels, and even relationships. Snoring, especially when paired with obstructive sleep apnea, has been linked to serious adverse health conditions such as heart disease.1 And it’s not just the snorer’s health that suffers. Approximately 20% of adults in the developed world consistently snore at night at volumes high enough to disturb their sleeping partners.2
So, when Cook was introduced to Zelegent, Inc. by M. Boyd Gillespie, MD, MSCR, chief of otolaryngology at the University of Tennessee Memphis and a principal investigator for Zelegent, Inc. an idea began to form.

“We saw a need for new options to help patients and their bed partners struggling with snoring,” said Thomas Cherry, global business leader for Cook’s Otolaryngology-Head & Neck Surgery (OHNS) specialty. “We found that Zelegent’s solution aligns perfectly with Cook’s mission to support minimally invasive procedures, and we are excited to help bring this technology to patients.”
Partnering with Zelegent would allow the OHNS team to expand the portfolio of products Cook could offer to physicians in the ear, nose, and throat (ENT) space and to enter the procedural realm of sleep medicine.
On January 8, Cook announced an agreement with Zelegent, Inc. in which the OHNS specialty would provide global sales, marketing, and distribution support to enhance treatment options for patients suffering from snoring. Then, on January 28, the two companies announced that one of these treatment options, the Elevo® Snoring Intervention Device, had received FDA clearance.

Product training begins
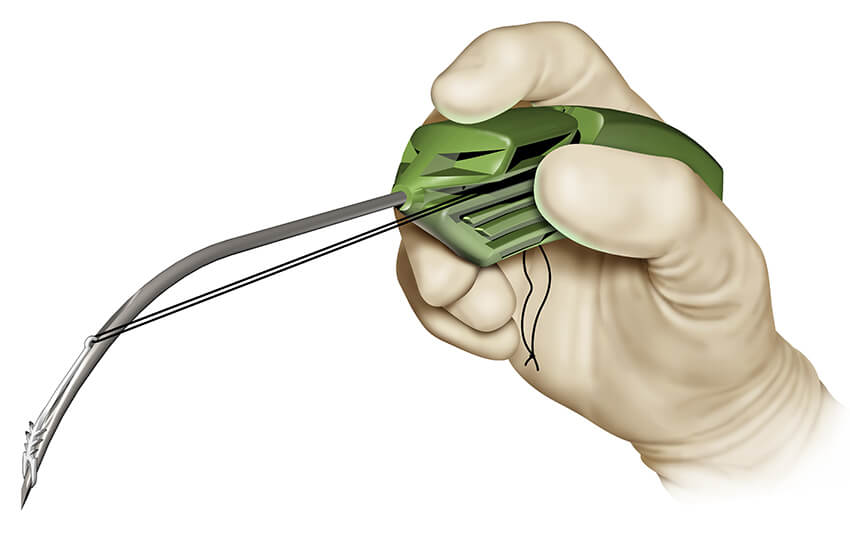
On January 31, Zelegent’s chief executive officer, Alexander Arrow, MD, came to Park 48 to teach OHNS district managers (DMs) about the history of Zelegent and Elevo and the Elevoplasty® procedure. Hands-on training was held in the form of a raw chicken lab in which the DMs were able to deploy the Elevo suture.
The delivery system for Elevo allows placement of specially shaped sutures that provide lift to the soft palate during the Elevoplasty procedure. The sutures are then naturally dissolved by the body. Elevoplasty can be performed under local anesthesia in an office setting without the need for surgery. Elevo is indicated for symptomatic, habitual, and social snoring due to palatal flutter.
“Elevoplasty is like a mini-face lift for the soft palate,” said Arrow. “We are honored to have the Elevo product adopted into a minimally invasive and innovative Cook product portfolio whose reputation dates back to Cook’s early work supporting pioneering physicians.”
“Patient-focused products like Elevo are the reason our team loves what we do, and this product training allowed us to get the product in our hands to see how our physicians will perform the procedure in-office,” said Drew Gidish, western regional sales manager for OHNS.
“The training was really interactive, which allowed us to ask Alex in-depth questions about the product and procedure so that we can better serve our customers in the field,” Drew added.
S.I.LE.N.C.E.
Zelegent recently completed a multi-center clinical trial designed to evaluate the safety and efficacy (effectiveness) of snoring intervention with Elevoplasty in an office-based setting. The Snoring Intervention via Elevoplasty in a Non-surgical Clinical Environment (S.I.LE.N.C.E.) clinical trial included both academic institutions and private practitioners. Included were several thought leaders on sleep disorder treatments and international lecturers on sleep surgery in the field of otolaryngology.
Prepare for launch
OHNS began a phased rollout in the US for Elevo at the beginning of the second quarter of 2019. Cook will be offering seven market access Vista® training courses in select cities, including Los Angeles, Chicago, and New York. The first Vista course took place on April 6 in Los Angeles.
“The sales team is really excited for the opportunity to bring this minimally invasive option to otolaryngologists to help them improve a patient’s quality of life,” Drew said.
Elevo will be displayed at several industry conferences throughout the year, most notably at the International Surgical Sleep Society’s annual meeting on May 9 and the American Academy of Otolaryngology-Head and Neck Surgery annual meeting on September 15.
A direct-to-consumer marketing campaign is planned for 2020.
“We’ve already seen a tremendous response from physicians and patients interested in Elevo as a result of our previous press releases,” Alex Brethauer, OHNS global product manager, said.
He added that the next step is to begin educating physicians on both Elevo and Elevoplasty by collaborating with physician subject-matter experts from the S.I.LE.N.C.E. clinical trial.
“We aim to deliver world-class education through a series of Vista educational events,” Alex said. “With a landmark clinical study to support the product and an underserviced patient population seeking a minimally invasive solution, we feel that Elevo could be a flagship product for OHNS and for Cook.”
For those people whose lives have been impacted by snoring, Elevo could have a substantial impact.
“Loud, long-term snoring can have countless negative side effects,” said Thomas. “This can be damaging to almost every aspect of a patient’s life. We’re very excited to provide a minimally invasive, office-based procedure to treat patients with this condition.”3


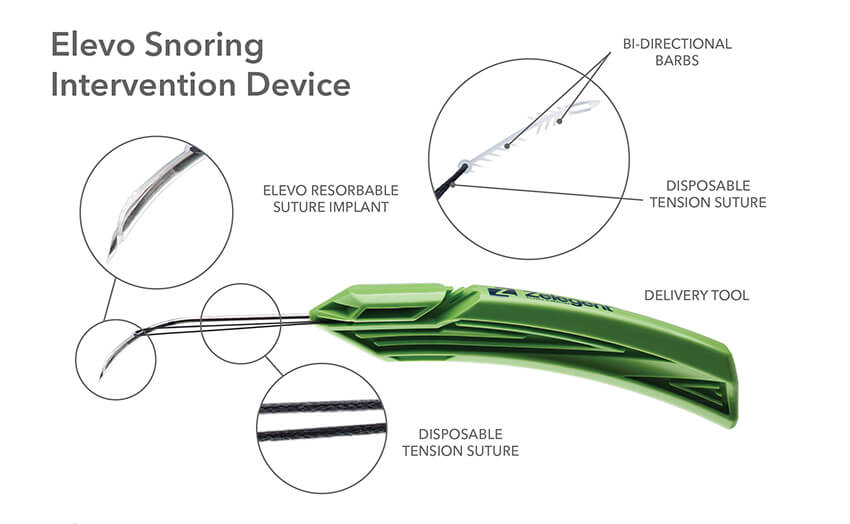
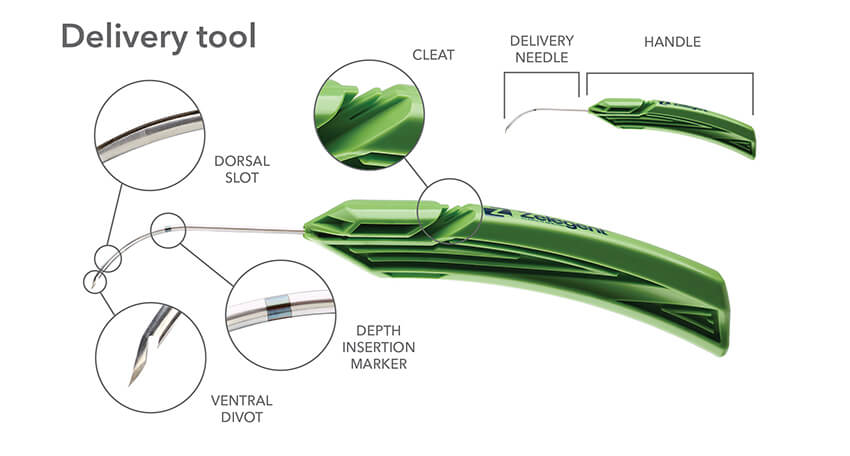
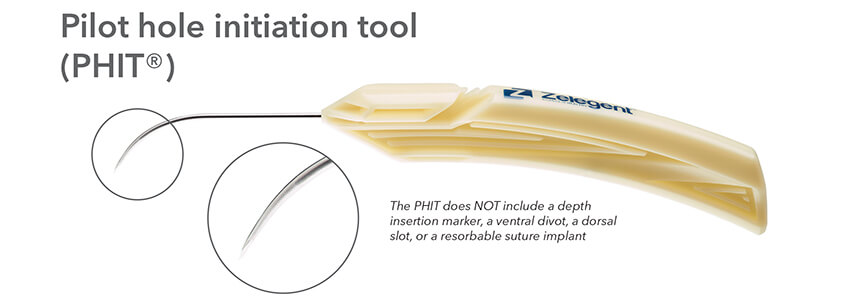
Indications for Use: The Elevo Kit Snoring Intervention Device is intended for use in stiffening the soft palate tissue, which may reduce the severity of snoring in some individuals.
Precautions: The Elevo Kit Snoring Intervention Device is not to be used in the treatment of obstructive sleep apnea.
Learn more
Term glossary
- Symptomatic snoring: Snoring that occurs when air flows over respiratory structures, such as the tongue, soft palate, and larynx, that have relaxed during sleep, causing them to vibrate.
- Habitual snoring: Snoring that occurs four or more nights a week.
- Social snoring: Snoring loud enough to disturb one’s bed partner during sleep.
- Palatal flutter: A fluttering motion in the soft palate of the mouth caused by tissue vibrations due to snoring.
For more information about Zelegent, Elevo, and Elevoplasty:
- Zelegent website: https://www.zelegent.com
- Elevo Vista website: https://vista.cookmedical.com/elevoplasty
- Elevo page: https://www.cookmedical.com/products/205b8629-8d05-4388-be18-4b359b3a5feb/
- Cook Medical and Zelegent, Inc. begin sales and marketing agreement: : https://www.cookmedical.com/newsroom/cook-medical-and-zelegent-agreement/
- Cook Medical collaboration with Zelegent, Inc. receives FDA clearance for snoring product: https://www.cookmedical.com/otolaryngology/cook-medical-collaboration-with-zelegent-inc-receives-fda-clearance-for-snoring-product/
—
1. Kapur VK. Obstructive sleep apnea: diagnosis, epidemiology, and economics. Respir Care. 2010;55(9):1155-1167.
2. Rubenfire M. How chronic snoring can cause heart disease. Michigan Health Web site. https://healthblog.uofmhealth.org/heart-health/how-chronic-snoring-can-cause-heart-disease. Published February 6, 2017. Accessed April 5, 2019.
3. S.I.LE.N.C.E. Clinical Study. Zelegent, Inc. zelegent.com/elevoplasty/silence-clinical-study/. Accessed April 18, 2019.
That’s one small step for Cook/Zelegent, one giant leap for mankind – bravo – I would like one myself…
Please keep us updated, I am sure that there is a need for this product in most homes. I would be interested in this device myself.
Thank you.
Keep us posted. I am sure my wife will be signing me up when it is available locally!
This is great…..now maybe we can cure the common cold. These problems have been around for a lifetime. I knew there was an answer out there somewhere. Boy am I happy the name COOK is tied to it, thank you what an interesting article.